Executive summary
Positive Results in the Half Year to
- 19% increase in revenues from clinical demand for SCENESSE® (afamelanotide 16mg).
- 94% increase in NPAT; 67% increase in EBIT.
- Net assets increased by 11%; 16% growth in cash to
$140.7 million since the start of the reporting period.
Increases year on year compared to six months to 31/12/21, unless stated otherwise.
All figures reported in Australian dollars, $.
| Change | ||||
| Revenues, $ | 29,355,042 | 24,631,266 | +19.2% | |
| Profit after tax, $ | 11,387,665 | 5,870,380 | +93.9% | |
| Basic Earnings per share, $ | 0.230 | 0.119 | +93.3% | |
| Cash, $ | 140,703,376 | +15.8%* | ||
*Increase from
For the six months ended
“Today’s excellent figures are the result of years of focus and financial discipline, while operationally facilitating supply of SCENESSE® to patients in
“The number of prescriptions for, patients receiving, and expert centres administering the therapy have all increased over the period.
“The performance exceeds our expectations, thereby precipitating the financial basis to expand our portfolio of melanocortins, diversify markets, and become a self-sufficient company.
“Working within our set budgets, we are now focused on bettering our results for the financial year end. It is an exciting moment in the history of the Company.”
Alongside the revenue result, CLINUVEL contained its expenses during the half year period to achieve a negligible rise of 1%, furthering its balance sheet. As of 31 December, CLINUVEL held cash and cash equivalents of over
“We have actively managed CLINUVEL’s commercial programs, translating to a steady increase in liquid assets, as well as earnings per share,”
“In parallel, we have made necessary investments to advance clinical research and new product development, while placing emphasis on cost control. There is not much debate around prudent fiscal management in a climate of macroeconomic uncertainty, therefore we keep a balance between strengthening the Group’s commercial basis and expansion. Most of all, I am pleased for patients and our shareholders, as our financial record and stability provides options for pursuing our long-term objectives.”
Financial performance
The 67% increase in net profit before tax was the fourteenth consecutive half year profit since the commencement of CLINUVEL’s commercial operations in
CLINUVEL is on track to remain within its projected overall expenditures of
The Company maintained a balance sheet free from external borrowings, with a rise in total assets of 11.4% to
CLINUVEL’s Appendix 4D Half Yearly Report is available on the Company’s website, www.clinuvel.com.
About
CLINUVEL (ASX: CUV;
CLINUVEL’s lead therapy, SCENESSE® (afamelanotide 16mg), is approved for commercial distribution in
SCENESSE®, PRÉNUMBRA®, and NEURACTHEL® are registered trademarks of CLINUVEL.
Authorised for ASX release by the Board of Directors of
Media Enquiries
Mr
Head of Investor Relations
Mr
Investor Enquiries
https://www.clinuvel.com/investors/contact-us
Forward-Looking Statements
This release contains forward-looking statements, which reflect the current beliefs and expectations of CLINUVEL’s management. Please see the full disclaimer on CLINUVEL’s website.
www.clinuvel.com
Level 11,
A photo accompanying this announcement is available at https://www.globenewswire.com/NewsRoom/AttachmentNg/5b11aca8-a262-48df-8297-ddb65974ca1c


CLINUVEL Financial Performance
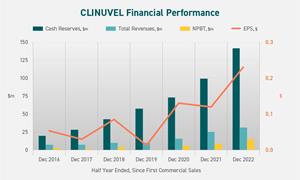
CLINUVEL Group financial performance for the half year ended 30 December, since first commercial sales.
2023 GlobeNewswire, Inc., source


